In this article we will discuss about:- 1. Definition of Biosurfactants 2. Classification of Biosurfactants 3. Enzyme Synthesized 4. Microbial Biosurfactants 5. Production 6. Applications.
Contents:
- Definition of Biosurfactants
- Classification of Biosurfactants
- Enzyme Synthesized Biosurfactants
- Microbial Biosurfactants
- Production of Biosurfactant
- Applications of Biosurfactants
1. Definition of Biosurfactants:
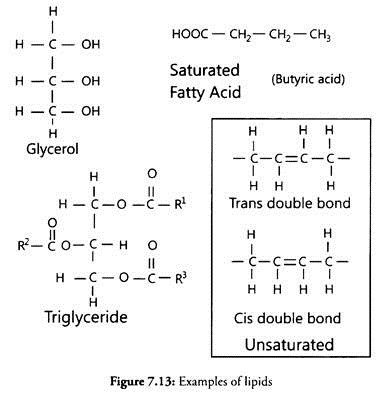
Biosurfactants can be defined as amphipathic molecules with both hydrophilic and hydrophobic (generally hydro carbons) moieties that partition preferentially at the interface between fluid phases with different degrees of polarity and hydrogen bonding such as oil/water or air/water interfaces. This allows the monomers to form micelles or to aggregate into micellar tubes, micellar bilayers and vesicles (Fig. 14.1).
Surfactants are also characterized by an ability to enhance the formation of emulsions of immiscible liquids such as hydrocarbons and water (Fig. 14.2).
These properties render surfactants capable of reducing surface and interfacial tension and forming micro-elusion where hydrocarbons can solubilize in hydrocarbons. These characteristics confer excellent detergency, emulsifying, foaming and dispensing traits. They readily form foam. Bio-surfactants can be categorized on the energy charge associated with hydrophilic group in the non-ionic, anionic, cationic or amphoteric. These have a great utility as wetting agents and for their effectiveness as emulsifier.
Bio-surfactants at present have 9.4 billion dollars sales only in USA and is likely to increase at a rate of 3-5% annually. At present surfactants are in use chemicals derived from petroleum.
But in recent times microbial surfactants are receiving increased demand due to the following features:
(1) Their diversity and environment friendly nature,
(2) Possibility of their production through fermentation,
(3) Their potential applications in the environmental protection,
(4) Crude oil recovery,
(5) They are useful in health care and food processing.
Bio-surfactants are superior to chemical surfactants due to the following features:
(1) They have lower toxicity,
(2) They are easily biodegradable,
(3) Better environmental compatibility,
(4) Higher foaming ability,
(5) Higher selectivity and specific activity at extreme temperature, pH and salinity.
(6) They can be synthesized from renewable feed stocks.
Methods of microorganisms screening for their bio-surfactant production potential are:
(a) Axiometric drop shape analysis.
(b) Colorimetric estimation (anionic surfactants react with cationic indicator to form coloured complex).
(c) A rayeid drop collapsing test.
(d) Direct TLC method.
(e) Estimation of the emulsification index value.
2. Classification of Biosurfactants
:
According to their Polarity, chemical composition and microbial origin biosurfactants can be classified into:
(1) Glycolipids,
(2) Lipopeptides or lipoprotein,
(3) Phospholipids and fatty acids (mycolic acids),
(4) Polysaccharide-lipid complexes,
(5) Complete cell surface,
(6) Polymeric surfactants,
(7) The particulate type.
(i) Biosurfactants can also be classified according to their source of production (Fig. 14.3).
(ii) Biosurfactants can be classified based on chemical composition and microbial origin as:
(1) Hydrophilic moiety containing amino acids or polypeptides anions or cations (mono, di or polysaccharides),
(2) Hydrophobic moiety containing unsaturated, saturated fatty acids
a. Lipopeptides
b. Gramicidins (B.brevis)
c. Fatty acid, phospholipids, and
d. Polymeric.
(iii) Biosurfactants are classified on the basis of producing source, as:
(a) Microbial biosurfactants, and
(b) Enzymatic synthesized surfactants
(iv) Classification by Type of Substrate Used:
1. Those producing biosurfactants with alkanes as carbon sources (Corynebacterium sp and Arthrobacterium sp)
2. Those producing biosurfactants with water soluble substrates as carbon sources (Bacillus sp)
3. Those producing biosurfactants with alkanes and water soluble substrates as carbon sources (Pseudomonas sp)
3. Enzyme Synthesized Biosurfactants
:
Many isolated enzymes that catalyze hydrolysis, alcoholysis, condensation, acylation or esterification reactions have been used for the production of various surfactants including
monoglycerides, phospholipids, glycolipids and amino acids based surfactants from relatively inexpensive raw materials such as fats and plant oils. Enzymatic methods of synthesis of surfactants have several advantages over conventional chemical synthesis.
The enzymatic methods have low energy requirement, minimal thermal degradation, high biodegradability and radioactive immobilized microbial lipases such as lipozyme from Rhizomucer miehei or lipase sp 435 from Candida antarctica are the most frequently used enzymes.
Using this technology Glycolipids such as fructose oleate, monocapryloyl-1 D-fructofuronase, B- 1-1fructopyrasanose-oleate, a mixture of C-1 and C-6 monopalmintoyl fructose, 6-octano glucose and various primary monoesters of sugar alcohols such as sorbitol, mannitol and xylitol have been produced.
Aliphatic alcohol glycosides, phenol glycosides, vitamin glycosides, glycerol glycolipids and glycolipid biosurfactant, fructose monooleates, monoole aleoyl glycerol and uncommon hydroxyl compounds such as 2-hydroxy ethyl-trimethyl silane which is attached to C1 glucose are among the recently synthesized enzymatic surfactants. Major characteristic features of microbial and enzyme synthesized surfactants are precised in Table 14.1.
However, disadvantages of enzyme technology together with non-aqueous phase kinetics will immensely contribute towards the elucidation of these issues. Similarly main disadvantage of high recovery costs and large liquid waste volumes are likely to be eliminated as a result of future research in strain development, metabolic engineering and fermentation technology.
4. Microbial Biosurfactants
:
Some of the important classes of biosurfactants are listed along with producing microorganism in table 14.2 and described below:
1. Glycolipids:
These are carbohydrates in combination with long chain aliphatic acids or hydroxyaliphatic acids. The linkage is by means of either ether or an ester group. Rhamnolipids are biosurfactants in which one or two molecules of rhannonose linked to one or two molecules of β-hydroxy decanic acid (Fig. 14.4). These are produced by Pseudomonas aeruginosa.
Trehalolipids are other group of biosurfactants. Species of Mycobacterium, Nocarchia and Corynebacterium are reported to produce these biosurfactants which include disaccharide trehalose linked at C6 and C6 associated with mycolic acid (Fig. 14.5).
Mycolic acids are long chain and branched β-hydroxy fatty acids. Rhodococcus erythropolis and Arthrobacter sp are reported to elaborate with lowered surface and interfacial tension in culture.
Sophorolipids are other category of glycolipids which are mainly elaborated by yeasts such as Torulopsis bombicola, T.petrophilum and T.apicola in which sophore is linked to a long chain hydroxy fatty acid by glycosodic linkage consisting of atleast 6-8 different hydrophobic sophorolipids (Fig. 14.6).
Glucose lipids contain 3-hydroxy fatty acids Rubiwettin RG1 (Fig. 14.7) and one produced by the marine bacterial strain MM1 are two important glucose lipids. Rubiwettin RG1 lowers from strain tension of saline to 26 mN/m.
2. Lipopeptides and Lipoproteins:
In these biosurfactants lipids are attached to a polypeptide chain. Surfactin which is produced by Bacillus subtilis ATCC21332 is a seven amino acid ring structure coupled to a fatty acid chain via lactone linkage (Fig. 14.7). Its surface tension is low (27.9 mN/m), at concentration as low as 0.005%. Lichenysin is another Lipopeptide biosurfactant elaborated by Bacillus lichenifermin. This biosurfactant lowers surface tension of water to 27 mN/m and the interfacial tension between water and n-hexadecane to 0.36 mN/m.
3. Fatty Acids, Phospholipids and Neutral Lipids:
Several yeasts and bacteria such as Acinetobacter sp produces phosphatidyl ethanolamine rich vesicles which form optically clear micro emulsions of alkanes in water.
4. Particulate Biosurfactants:
Extracellular membrane vesicles partition hydrocarbons to form a micro-emulsion which plays an important role in alkane uptake by microbial cells. Vesicles of Acinetobacter sp with a diameter of 20-50 nm and a buoyant density of 1.158 cubic g/cm and composed of protein phospholipids and lipopolysaccharides.
5. Surface Active Antibiotics:
Gramicidins, polymyxins and antibiotic TA comes under this category and they are elaborated by Brevibacterium brevi, B. polymxya and Myxococcus xáanthus respectively. Gramicidin S is cyclosymmetric decapeptide and has antibacterial activity which is attributed to its high surface activity.
Polymyxin D is a decapeptide in which amino acids 3 through 10 form a cyclic octapeptide A branched chain fatty acid is connected to the terminal 2,4-diaminobutyric acid (DAB). Polymixins are able to solubilize certain membrane enzymes. Antibiotic TA inhibits peptidoglycan synthesis by interfering with polymerization of the lipid disaccharide pentapeptide. It has interesting chemotherapeutic applications.
6. Polymeric Microbial Surfactants:
Most of these polymeric biosurfactants are heterosaccharide containing proteins. The best studied polymeric biosurfactants are emulsan, liposan, alasan and lipomanans produced by Acinetobacter calcoacetices. Emulsion is an effective emulsifying agent for hydrocarbon in water even at a concentration as low as 0.001 to 0.01 percent. Liposan produced by Candida lipolytica is an extracellular water soluble emulsifier composed of 83% carbohydrate and 17% protein.
5. Production of Biosurfactant:
Though the biosurfactants are produced by hydrocarbon degrading bacteria, they are readily produced on water soluble compounds such as glucose, sucrose, glycerol or ethanol. Biosurfactant production are either growth associated, growth limiting condition or resting or immobilized cells or they are produced with addition of precursors. Generally their production is influenced by the carbon source, nitrogen, phosphate source and other environmental factors. List of microorganisms producing different types of biosurfactant are listed in table 14.3.
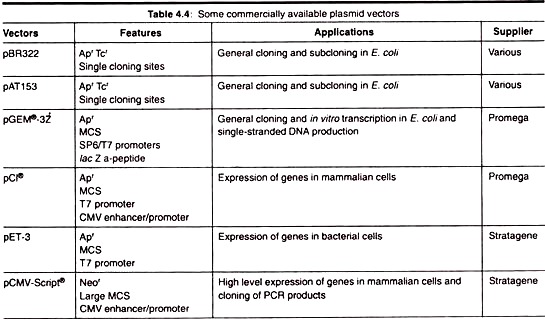
Different methods of biosurfactants production are precised in Table 14.4.
Culture conditions such as pH, temperature, dissolved oxygen and ionic strength also influence biosurfactant production. Temperature is especially important in the case of Arthrobacter paraineus ATTCC 1958, Rhizomucor erythropolis and Pseudomonas sp Bacillus subtilis CO2 is reported to produce three fold more lipopeptide under anaerobic condition when compared to aerobic condition. Carbon, nitrogen and phosphate sources, metal ions and other additives used in media formulation may play a critical role in the production and yield of biosurfactant.
Carbon substrate is reported to act in an order to reduce production costs, use of cheaper substrates such as rice hull hydrolysate, starch waste liquors, domestic wastes, potato processing wastes and they has been recommended. Similarly use of olive oil mill effluents and peat pressate has also been recommended.
Fermentative Production:
Since biosurfactants are diverse group of compounds produced by a variety of microbial species, it is difficult to develop general guidelines for process development. Although, most biosurfactants are released into the culture medium throughout the exponential phase, some can also be produced by resting cells or by immobilized biocatalysts.
The production of biosurfactants can be carried out using batch or continuous fermentation. The production of biosurfactants that can be produced by resting cells such as glycolipids by Torulopsis bombicola can be carried out by immobilized biocatalyst. Its production can be increased by online removal using an adsorption column or by foam fractionation, application of air lift fermenter and aqueous two phase fermentation.
Critical Role of Carbon Source in Biosurfactant Production:
These carbon sources are used either singly or in combination and produce glycolipids as shown in Fig. 14.9 and Table 14.5.
Solid state fermentation using soya bean, curd residue using a recombinant B subtilis has been reported to yield four times higher than that in submerged fermentation. Multi-organism strategy for biosurfactant production such as microbial single oil is obtained from Lipomyces or Chlorella. The single cell oil obtained from microorganism is used as a substratum for production of glycolipid by Candida bombicola. Similarly sophorose lipids are produced by two stage fed batch process using C bombicola and Cryptococcus curvatus. Some of the cheaper raw materials employed in biosurfactant production are listed in Table 14.6.
The separation and concentration of biosurfactants can be achieved by a few steps such as precipitation, organic extraction and absorption chromatography. Ultrafiltration can also be used to recover biosurfactant from fermentation broth. General methods of downstream process for isolation and purification of biosurfactants are precised in table 14.7.
6. Applications of Biosurfactants:
Biosurfactants are used in variety fields and have wide range of applications (Table 14.8).
In view of ecofriendly nature they have received great interest in the recent past in their production. The market is extremely competitive and there is a need to develop cost effective process of production of high yield and activity. Microorganisms of extremophiles nature and genetically engineered bacteria are need of the hour.
As we further understand both the applications, limitations of biosurfactants and their specific properties such as emulsification and demulsification foaming; water binding capacity spreading and wetting properties, viscosity and consistency will likely find application in an increasingly broad range of industrial, household processes and products.
A further understanding of the genetics and biochemistry of biosurfactant synthesis will make possible more effective in situ and ex situ production, application and assessment. As new biosurfactants continue to be discovered and existing ones become better characterized their utility is likely to expand dramatically through a multitude of specialized applications optimized conditions and modified processes.